How Does Soap Clean Clothes?
Detergency: The Science of How Soap Cleans Clothes
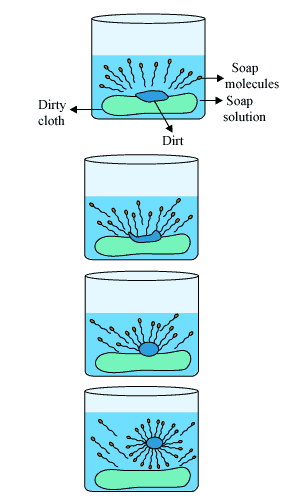
Soap cleans clothes through a process called detergency—a combination of reduced water surface tension, the “roll-up” mechanism that lifts oils and dirt from fabric fibers, and electrostatic repulsion that keeps removed soils suspended in wash water rather than redepositing onto cleaned surfaces. Soap molecules have a hydrophilic (water-seeking) head and a hydrophobic (water-repelling) tail, which allows them to simultaneously interact with water and oily substances, binding to dirt particles and rinsing them away.
A detergent must possess three essential properties: good wetting ability to make contact with fabric, soil removal capability, and the ability to keep removed dirt suspended so it cannot redeposit. These principles apply to all soap and synthetic surfactants used in laundry cleaning.
Surface Tension and Soap’s Role
Pure water has a surface tension of 72.8 mN/m (approximately 72.8 dynes/cm) at 20°C. Soap is a surface-active compound that reduces this surface tension by a factor of three or more, lowering it to approximately 25 dynes/cm or lower. At the fabric-water and oil-water interfaces, the long alkyl chain of soap molecules tends to move away from the water phase, decreasing surface tension because the force acts opposite to the inward pull of water molecules.

The hydrophilic portion of a soap molecule is the carboxylate ion (RCO₂⁻), while the long alkaline chain is hydrophobic. This dual nature enables soap to interact with both water and oily substances simultaneously, making it an effective cleaning agent.
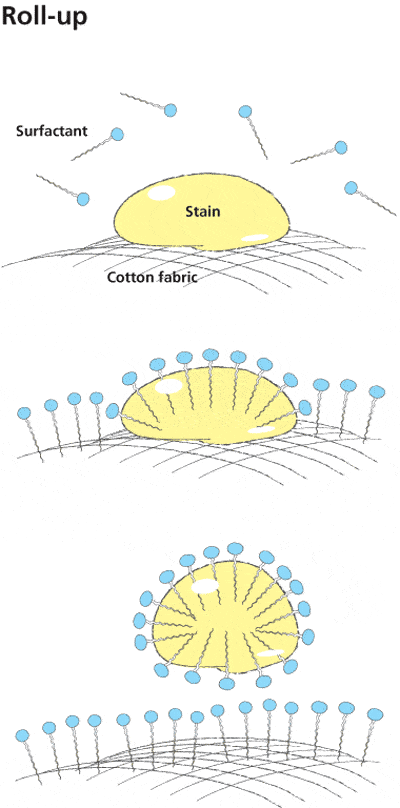
The Roll-Up Mechanism for Hydrophobic Soils
When considering an oil drop on fabric, the shape depends on three surface tension forces working in concert. The surface tension between the oil drop and the surrounding aqueous phase is represented as T_L. The surface tension between the fabric and the surrounding aqueous phase is T_S. The surface tension between the fabric and oil drop is represented as T_LS.
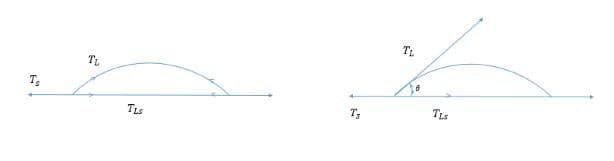
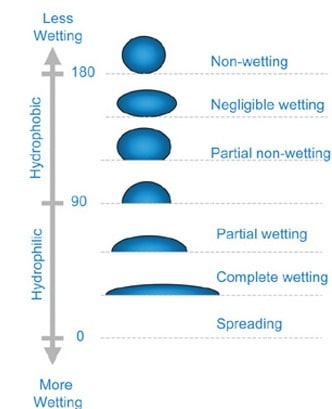
When T_LS plus T_L increases relative to T_S, the area of contact between oil and fiber decreases. This causes the oil to form a sphere with a decreasing contact angle (θ). Contact angles smaller than 90° indicate hydrophilic surfaces, while angles greater than 90° indicate hydrophobic surfaces. When the forces reach equilibrium, Young’s equation applies: γ_SG – γ_SL – γ_LG cos θ_C = 0, which relates interfacial energies to the contact angle.
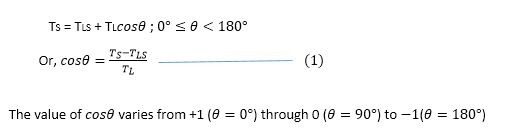
The removal of hydrophobic dirt occurs through a specific sequence. First, the hydrophobic tails of soap molecules saturate both the dirt or oil and the fabric surface. Second, fiber-water and oil-water surface tension decreases due to surfactant adsorption. Third, because the surface tension between fiber and oil remains unchanged, the cosine of the contact angle becomes negative, indicating the contact angle exceeds 90°. Fourth, the unchanged surface tension between fiber and oil combined with electrostatic repulsion between the negatively charged fiber-water and oil-water interfaces causes the oil droplet to reduce its surface area by rolling up into a sphere.
The oil droplet begins to lift from the fiber and is removed into the solution, where it remains suspended by electrostatic repulsion. The negatively charged repulsion between the oil-water and fiber-water interfaces prevents the dirt or oil from redepositing onto the fabric. This is why soap is effective at keeping soils suspended in wash water rather than allowing them to redeposit.
Micelle Formation and Critical Micelle Concentration
Soap molecules form micelles, which are tiny spherical aggregates with polar hydrophilic groups on the outside and lipophilic pockets on the inside. The critical micelle concentration (CMC) is the surfactant concentration at which micelles begin to form. Above the CMC, additional surfactant molecules simply increase the number of micelles rather than further reducing surface tension.
| Surfactant | Critical Micelle Concentration (CMC) |
|---|---|
| Sodium Dodecyl Sulfate | 0.0083 M |
| Sodium Octyl Sulfate | 0.13 M |
Soap solubilizes particles and grime by trapping oil and grease molecules inside the lipophilic core of micelles, effectively shielding them from water and allowing them to be washed away. Removal of oily soil occurs primarily through modification of contact angles and release of the oil in the form of an emulsion, not through solubilization alone.
Removal of Hydrophilic (Polar) Soils
Hydrophilic soils such as salts, sugars, and polar compounds are removed through a different mechanism. The hydrophilic head of the soap molecule orients toward the polar dirt particle. A second layer then forms where the hydrophobic tail orients itself toward the first layer of tails, creating a bilayer structure. This arrangement allows the polar soil to be lifted and suspended in the wash water.
Water Hardness and Soap Performance
Hard water contains calcium and magnesium ions that react with soap to form soap scum rather than producing lather. The reaction proceeds as follows: 2 RCO₂H + CaO → (RCO₂)₂Ca + H₂O, where R represents the fatty acid chain. Modern synthetic detergents include builders such as sodium tripolyphosphate or zeolites to soften water and improve cleaning performance in areas with hard water.
World production of surfactants reaches approximately 15 million tons annually, with linear alkylbenzene sulfonates accounting for 1.7 million tons per year, fatty alcohol ethoxylates for 700,000 tons per year, and alkylphenol ethoxylates for 500,000 tons per year. This industrial scale demonstrates the essential role surfactants play in cleaning and hygiene worldwide.
For more details on the chemistry of soap and micelle formation, read our guide on Soap and Micelle Properties.
References
- Wikipedia contributors. (2024). Surface Tension. Wikipedia.
- Wikipedia contributors. (2024). Critical Micelle Concentration. Wikipedia.
- Wikipedia contributors. (2024). Soap. Wikipedia.
- Wikipedia contributors. (2024). Surfactant. Wikipedia.
- Wikipedia contributors. (2024). Contact Angle. Wikipedia.
- Trotman, E.R. (1984). Dyeing and Chemical Technology of Textile Fibres. Charles Griffin & Company.
- Roy Choudhury, A.K. (2006). Textile Preparation and Dyeing. Science Publishers.

