What is Soap – Properties of Soap and Its Critical Micelle Concentration
Soap
Soaps are metallic salts of fatty acids (saturated or unsaturated) containing from 8 to 22 carbon atoms. It’s a natural cleansing agent. There may be various kinds of metallic salt but sodium and potassium salts are used as detergents.
Commercially soap is produced by boiling natural fats/oils with aqueous solution of sodium or potassium hydroxide. This reaction is called Saponification.
The potassium soaps tend to be softer and more soluble in water than the corresponding sodium soaps
Chemistry of Soap Manufacturing
Fat Saponification Process
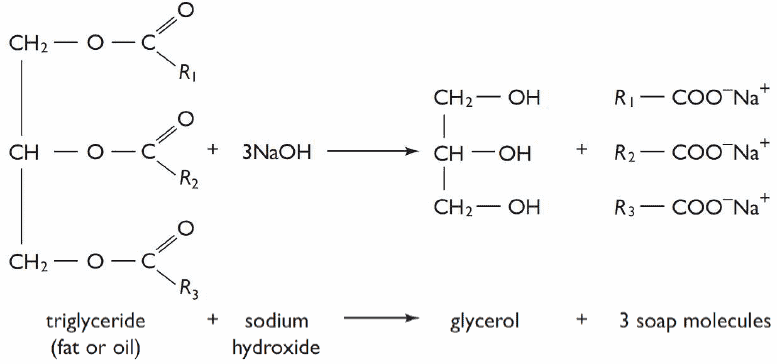
Sodium soaps are sparingly soluble in strong sodium chloride solution. The mixture of soap and glycerol in aqueous solution obtained by saponification is saturated with common salt where the soap is precipitated. As soap is lighter than glycerol it’ll rise to the surface. Then it’s skimmed off i.e. the best parts from the surface is removed.
To remove excess alkali or salt solution it’s washed with cold water. Then it’s cast into cakes/bars and dried.
The liquor is evaporated under reduced pressure and glycerol is recovered by distillation
Fatty Acid Neutralization Process
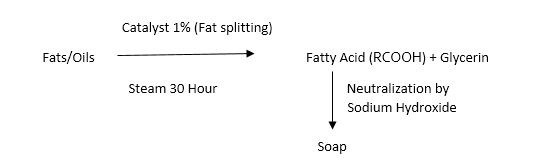
(In this process alkalinity can be controlled as we add in known concentrations)
Fatty Methyl Ester Process
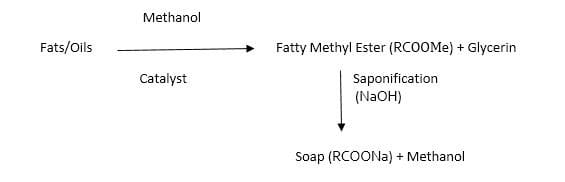
(Glycerin found in this process are more pure than other processes)
Functional Properties of Soap
Solubility
- An increase in the size of monovalent cation (base) increases solubility & an increase in the size of di/tri valent cation decreases solubility
- Carbon chain length of soap increases, solubility decreases but cleansing power increases.
- The presence of unsaturation results in an increase in solubility.
In general, with the increase of solubility the softness and mushiness property of soap also increases
Lathering and Cleansing Attributes
- The use of fatty acids of C-10 to C-12 chain length is most preferred for lathering attributes.
- The use of fatty acid of C-16 to C-18 chain length is most preferred for cleansing attributes.
The most suitable soap is made from the combination of coconut ( C-10 to C-12 ) and tallow( C-16 to C-18 ) from the view point of lathering and cleansing attributes .
The effectiveness of a soap solution as a detergent will be influenced to a marked degree by –
- Nature of the fatty acid
- Conditions of temperature
- Concentration in which it is to be used
Disadvantages
- Doesn’t work on hard water
- Doesn’t work on acidic solution
- Doesn’t work in the presence of sodium ion as excess sodium ion precipitates soap.
Micelle
As the concentration of surfactant in a solution is increased it becomes energetically favorable for the individual molecules, or monomers, to combine together to form large aggregates, or micelles, which shield the hydrophobic components from the solution.
It’s a group of surfactant molecules associated in a cluster.
Here, a stearate ion is represented –
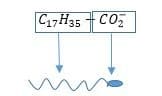
(The long alkaline chain is the hydrophobic and carboxylate ion is the hydrophilic part)
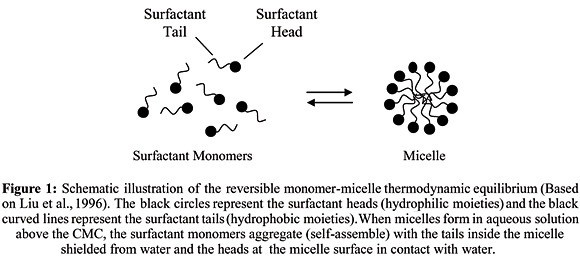
Structures of Micelle
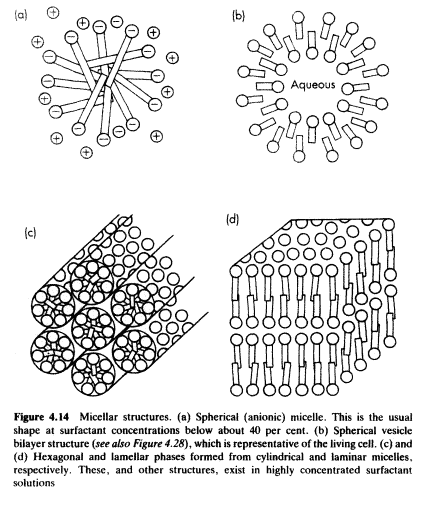
Only (a) & (d) is of our concern as we only find these two structures in surfactant solution.
CMC
The concentration above which micelle formation becomes appreciable is termed as the Critical micelle concentration (C.M.C.)
At low surfactant concentration the surfactant molecules arrange on the surface. When more surfactant is added the surface tension of the solution starts to rapidly decrease since more and more surfactant molecules will be on the surface.
When the surface becomes saturated, the addition of the surfactant molecules will lead to formation of micelles. This concentration point is called critical micelle concentration.
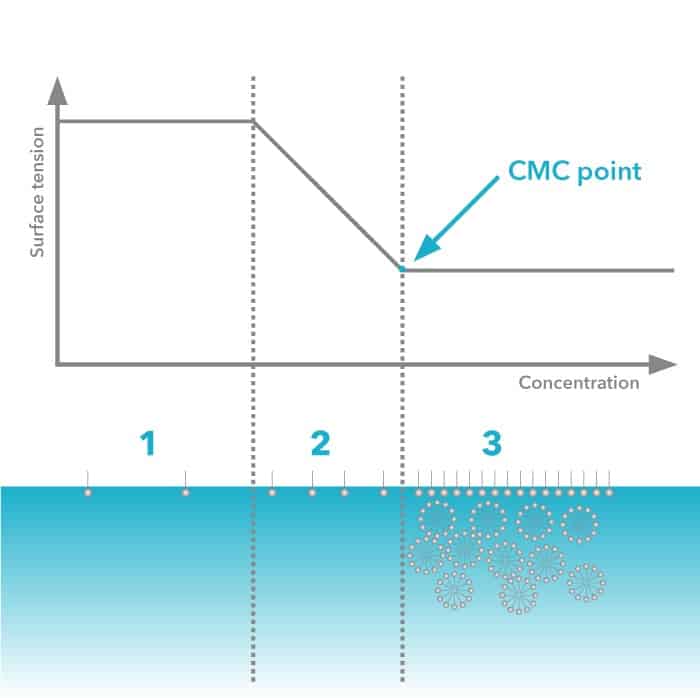
- At very low surfactant concentration only slight change in surface tension is detected.
- Addition of surfactant decreases the surface tension drastically
- At CMC point, surface becomes saturated and the addition of surfactant molecules do not effect on the surface tension.
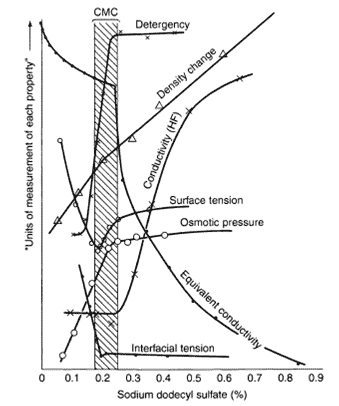
Factors Affecting CMC
No. of Carbon Atom
Increasing hydrophobic part (no. of carbon atom) will result in a decrease of c.m.c. In aqueous medium, the c.m.c. of ionic surfactants is approximately halved by the addition of each CH2 group .For non-ionic surfactants this effect is usually even more pronounced.
This trend usually continues up to about the C16member. Above the C18 member the c.m.c. tends to be approximately constant. This is probably the result of coiling of the long hydrocarbon chains in the water phase.

Thermal Agitation
Micelle formation is opposed by thermal agitation and c.m.c.’s would thus be expected to increase with increasing temperature but not always.
Addition of Electrolytes

With ionic micelles, the addition of simple electrolyte reduces the repulsion between the charged groups at the surface of the micelle. The c.m.c. is, therefore, lowered.
Addition of Organic Molecules
Organic molecules may influence c.m.c.’s at higher additive concentrations by virtue of their influence on water structuring. Sugars are structure-makers and as such cause a lowering of c.m.c., whereas urea and formamide are structure-breakers and their addition causes an increase in c.m.c.
Micelles containing more than one surfactant often form readily with a c.m.c. lower than any of the c.m.c.’s of the pure constituents.
References
Books:
Colloid and Surface Chemistry by Duncan J Shaw
Dyeing and Chemical Technology of Textile Fibres by E.R. Trotman
Lectures:
Md. Koushic Uddin
Lecturer, Dept. of Textile Engineering, Ahsanullah University of Science and Technology
Images:

