Why Are My Clothes Getting Bleach Spots?
What Causes Bleach Spots on Clothes?
If you’ve ever found a white or light-colored spot on a dark garment, you know the frustration of bleach spots. Whether it’s a single spot or an entire garment that’s been affected, these spots ruin clothes and catch you off guard. But what causes them?
Bleach spots on textile garments result from three primary sources: undiluted sodium hypochlorite bleach contacting fabric directly, residual bleach in washing machines from improper dilution, and chemical reactions between bleach and detergent ingredients. Household sodium hypochlorite solutions typically contain 3–6% active chlorine by volume, making them highly reactive with organic dyes in fabrics.
What Causes Bleach Spots on Clothes?

You expect your clothes to emerge clean and free of blemishes when you do laundry. But sometimes, bleach spots appear after washing. Sodium hypochlorite—the active ingredient in household chlorine bleach— oxidizes and destroys the dyes in textile fibers on contact. Even diluted solutions can cause spotting when poured too close to fabric or when inadequate water circulation leaves pools of bleach against the garment surface.
1. You Accidentally Spilled Bleach on Your Clothes
Pouring undiluted bleach directly onto fabric causes immediate oxidative damage to textile dyes. A single drop of full-strength sodium hypochlorite at 5% active chlorine concentration can destroy color in a 2–3 cm diameter area within seconds. If you spill bleach on your clothes, rinse the affected area immediately with cold running water for at least 10 minutes to flush the chemical before it penetrates the fiber.
2. Your Clothes Were Stored in a Place Where Bleach Fumes Were Present
Storing textile garments in enclosed spaces near open bleach containers or recently bleached items allows chlorine gas to settle onto fabric surfaces. While this typically causes generalized fading rather than distinct spots, concentrated fume exposure near the top of a storage bin can produce localized bleach spotting. Keep bleach containers sealed and store laundry areas separately from bleach storage.
3. You Used Too Much Bleach in the Wash

US regulations for food-processing sanitization allow solutions up to 200 parts per million (ppm) of available chlorine. For household laundry, the CDC recommends a 1-in-47 dilution of standard bleach (approximately 1 part bleach to 47 parts water) for effective sanitization. Exceeding this ratio—whether through measuring errors or adding undiluted bleach directly to the drum—creates solutions of 500–1,000 ppm or higher that oxidize textile dyes within the first 5 minutes of agitation.
4. The Bleach Was Old and Expired
Household sodium hypochlorite bleach degrades at a rate of approximately 15–20% loss in active chlorine concentration per year, even when sealed and stored at room temperature. After 12 months of storage, a fresh bottle labeled at 6% active chlorine may contain only 4–5% active ingredient. Degraded bleach produces inconsistent whitening results and can generate breakdown byproducts that react unpredictably with fabric dyes and detergent components.
5. A Chemical Reaction Occurred Between the Bleach and Another Substance in the Wash
Combining chlorine bleach with cleaning agents containing ammonia, acetic acid (white vinegar beyond small decorative amounts), or hydrogen peroxide triggers violent chemical reactions. Mixing bleach with ammonia-based cleaners releases chloramine gas, which settles on fabric surfaces and causes oxidative spotting. When bleach reacts with hydrogen peroxide (used in color-safe bleach alternatives), both agents decompose before either can clean effectively, and the resulting solution can be more damaging to textiles than bleach alone.
How to Restore Fabric with Bleach Stain

Bleach destroys textile dyes by oxidation—a permanent chemical change to the organic dye molecules in the fiber. Unlike soil removal, which lifts dirt from fabric, bleach stain restoration requires neutralizing residual bleach and then re-dyeing the affected area to restore the original color. Complete restoration is possible on most synthetic and cotton fabrics; natural protein fibers like silk and wool respond poorly to bleach damage and may not accept re-dyeing evenly.
Here’s What You’ll Need
- A small bowl or basin
- A clean white cloth (undyed to prevent color transfer)
- 1 tablespoon of baking soda (sodium bicarbonate)
- 1 cup of cold water
- 1/2 cup of white vinegar (5% acetic acid)
- A soft-bristled brush (old toothbrush works well)
- Fabric dye matching the original garment color
Step-by-Step Guide
The restoration process follows a clear sequence: soak in cold water, apply baking soda paste to neutralize residual bleach, treat with white vinegar to balance pH, rinse thoroughly, and finally re-dye the affected area. Each step takes approximately 15 minutes. For best results, complete all steps within 24 hours of the bleach incident—older bleach damage penetrates deeper into the fiber structure.
Step 1: Soak the affected area of the textile in cold water for 15 minutes. Cold water (10–15°C) prevents the bleach from penetrating deeper into the fiber while loosening surface residue. Do not use warm or hot water, as elevated temperatures accelerate oxidative degradation of textile fibers.
Step 2: Mix 1 tablespoon of baking soda with enough cold water to form a thick paste (approximately 3 parts baking soda to 1 part water by volume). Apply the paste directly to each bleach spot and allow it to sit for 15 minutes. Baking soda (sodium bicarbonate) neutralizes chlorine bleach through an acid-base reaction that produces sodium chloride, water, and carbon dioxide.

Step 3: Using the soft-bristled brush, work the baking soda paste into the bleach spots using gentle circular motions for 2–3 minutes. Rinse the area thoroughly with cold water. Inspect the spots—severe oxidation may leave the fibers permanently lightened despite neutralization.
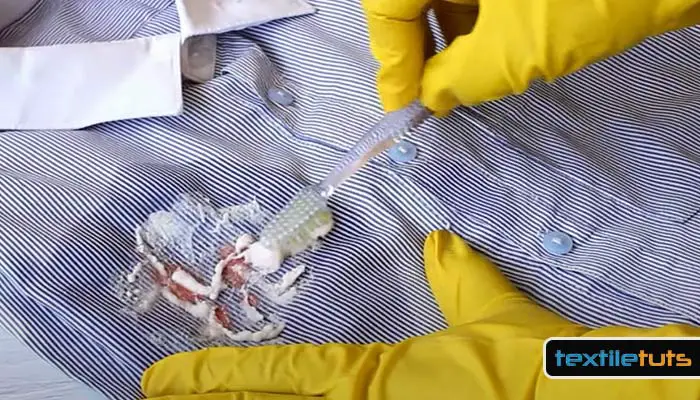
Step 4: Prepare a solution of 1 part white vinegar (5% acetic acid) to 2 parts cold water. Soak the affected area for 15 minutes. The acetic acid in white vinegar neutralizes any remaining alkaline residue from the baking soda treatment and helps prepare the fabric fiber to accept the dye evenly. Rinse the area with cold water after soaking.

Step 5: Rinse the area thoroughly with cold water until the water runs completely clear. Then wash the garment as usual using laundry detergent at the manufacturer-recommended dose. If bleach spots remain visible after drying, repeat the entire process once more before proceeding to re-dyeing.

Once the fabric is fully neutralized, apply a fabric dye that matches the original garment color. For synthetic fabrics (polyester, nylon, acrylic), use a dye specifically formulated for synthetic fibers—the dye must heat to at least 60°C (140°F) to penetrate the tightly packed polymer chains. For cotton and other cellulosic textiles, fiber-reactive dyes provide the most durable bond. Follow the dye manufacturer’s instructions precisely for best results.
How to Prevent Bleach Spots on Clothes
Preventing bleach spots is significantly more effective than attempting restoration. Sodium hypochlorite damage to textile dyes is irreversible on protein fibers and difficult to fully reverse on cellulosic and synthetic fabrics. Four evidence-based prevention methods address the most common causes of bleach spotting in household laundry.
Use a Laundry Bag

A mesh laundry bag (also called a garment bag) creates a physical barrier between delicate textiles and the aggressive bleach solution in the wash drum. Use a laundry bag with mesh openings of 2–3 mm for standard protection, or a fine-weave bag with 1-mm openings for heavily soiled loads requiring high bleach doses. Place the bag in the drum before adding detergent and bleach—never add garments to a machine that already contains undiluted bleach.
Be Careful When Pouring Bleach

Always pour bleach directly into the washing machine’s bleach dispenser drawer or the main wash drum before adding any fabric items. Never pour bleach directly onto clothing or into a basin where garments are already present. Hold the bleach bottle at a height of no more than 5 cm above the water surface when pouring, and use the bottle’s handle to maintain a secure grip throughout pouring.
Add Bleach to the Machine First

For top-loading and heavy-duty washing machines, add the measured bleach dose to the empty drum before loading laundry. Allow the machine to fill with water and run the agitation cycle for 2–3 minutes to fully dilute the bleach before adding garments. For front-loading machines with a pre-mix dispenser, add bleach to the designated compartment labeled “bleach” or “chlorine”—these dispensers mix bleach with water before releasing it into the drum.
Use Cold Water

Bleach reactivity increases exponentially with water temperature. At 40°C (104°F), sodium hypochlorite reacts with textile dyes approximately 4 times faster than at 20°C (68°F). Using cold water (15–20°C / 59–68°F) for bleach laundry loads provides adequate cleaning action while minimizing oxidative damage risk from localized hot spots in the wash drum. The enzyme-based stain removers in modern laundry detergents work optimally in cold water, providing complementary soil removal without requiring hot water.
Rinse Thoroughly

After completing any bleach wash cycle, run a minimum of two additional cold rinse cycles to flush residual sodium hypochlorite from the drum and fabric. The first rinse removes approximately 60–70% of residual bleach; the second removes an additional 25–30%. For heavily bleached loads (white linens, hospital laundry), a third rinse ensures total chlorine removal. Test the final rinse water with a chlorine test strip—water should register 0 ppm available chlorine before the final spin cycle.
Frequently Asked Questions
Understanding bleach chemistry helps you make informed decisions about textile care and restoration. These are the most common questions about bleach spots on clothes and their answers based on textile chemistry principles.
Do bleach spots go away?
Bleach spots do not disappear on their own. Sodium hypochlorite permanently oxidizes the organic dye molecules in textile fibers—the chemical change is irreversible through washing alone. The spot remains visible because the dye has been destroyed, not because soil remains. To eliminate the appearance of bleach spots, you must restore color to the affected area using fabric dye, color-restoration markers designed for textiles, or by reweaving matching threads into the damaged section.
What neutralizes bleach on clothing?
Sodium metabisulfite (Na₂S₂O₅) is the most effective commercial bleach neutralizer for textile applications, used at concentrations of 1–5 grams per liter of water for complete chlorine neutralization. For household use, sodium thiosulfate (“photographer’s hypo”) works at a ratio of approximately 1 teaspoon per liter of water. At home, you can use a paste of baking soda (sodium bicarbonate) followed by white vinegar (acetic acid) to neutralize surface bleach residue—the resulting chemical reaction produces carbon dioxide, water, and sodium acetate, none of which damage fabric.
Does vinegar remove bleach stains from clothes?
White vinegar does not remove bleach stains—it neutralizes residual bleach alkali after the oxidative damage has already occurred. Vinegar (5% acetic acid) reacts with residual sodium hypochlorite and any leftover baking soda to produce harmless byproducts. However, vinegar cannot reverse the oxidation of dye molecules in the fiber. The stain appearance remains—the vinegar merely prepares the fabric surface for effective re-dyeing by creating a pH-neutral foundation.
Can bleach ruin the fabric?
Bleach causes permanent structural damage to natural protein fibers including wool, silk, cashmere, and mohair. The disulfide bonds in keratin—the protein that forms wool and silk fibers—are cleaved by sodium hypochlorite, leading to fiber weakening, tissue-like degradation, and eventual shredding with minimal handling. Cotton and linen (cellulosic fibers) tolerate dilute bleach better but suffer progressive strength loss with repeated exposure. Synthetic fibers (polyester, nylon, acrylic) are the most bleach-resistant, though extended exposure to high concentrations causes fiber embrittlement and surface pitting.
Is Bleach the Cause of My Clothes Getting Bleach Spots or Could It Be My Detergent?
While detergent stains on clothes are a genuine concern, bleach remains the primary cause of the distinctive white spotting pattern characteristic of oxidative dye destruction. Detergent residue typically produces a bluish-gray film rather than sharp white spots, and it affects the entire garment surface rather than localized areas. However, certain detergent ingredients—notably optical brightening agents (OBAs) and phosphates—can interact with chlorine bleach to form compounds that accelerate dye fading. Check your detergent ingredients and avoid products containing both optical brighteners and chlorine bleach in the same wash load when laundering dark-colored textiles.
Final Words
Bleach spots result from the oxidative destruction of textile dyes by sodium hypochlorite—an irreversible chemical reaction that cannot be undone by washing alone. Understanding the five common causes of bleach spotting, following the step-by-step neutralization and restoration process, and applying evidence-based prevention methods will protect your wardrobe from preventable textile damage.
Many people discard garments after discovering bleach stains because they don’t realize restoration is possible. When bleach spots appear, neutralize the area immediately using the baking soda and white vinegar method, then re-dye with a color-matched fabric dye. Prevention—adding bleach before garments, using cold water, rinsing thoroughly, and using laundry bags for delicate items—eliminates the vast majority of bleach spotting incidents in household laundry.
References
- Bleichmittel. (2024). In Wikipedia, The Free Encyclopedia. Wikimedia Foundation. https://en.wikipedia.org/wiki/Bleaching
- American Association of Textile Chemists and Colorists (AATCC). (2024). AATCC Test Methods for Fabric Care and Bleach Stability. AATCC. https://www.aaatcc.org/
- U.S. Food and Drug Administration. (2024). Code of Federal Regulations, Title 21, Part 178 — Sanitizing Solutions. US Government Publishing Office. https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfcfr/CFRSearch.cfm?fr=178.1010
- Cotton Incorporated. (2024). Fabric Care: Bleach Guidelines for Textile Products. Cotton Works. https://www.cottonworks.com/en/topics/fabric-care/bleach/
- Centers for Disease Control and Prevention (CDC). (2024). Dilution Ratios for Bleach Solutions in Household Sanitation. US Department of Health and Human Services. https://www.cdc.gov/infection-control/hospital-air/hydrogen-peroxide.html
- U.S. Environmental Protection Agency (EPA). (2024). Safer Bleach Products and Textile Care Standards. EPA. https://www.epa.gov/

