Reactive Dyes – Classification | Properties | Dyeing Method
What is Reactive Dye?
What is Reactive Dye?
Reactive dye is a class of dye that forms a covalent bond with fiber molecules, becoming an integral part of the fiber itself. Unlike direct dyes or sulfur dyes that rely on physical adsorption, reactive dyes chemically bond to the terminal –OH groups of cellulosic fibers (cotton, rayon, linen) and the –NH₂ groups of polyamide and wool fibers. This covalent bond is what gives reactive dyes their hallmark advantage: permanent coloration that withstands repeated laundering without bleeding or fading. The reactive dye structure S–F–T–X breaks down as: S (chromophore, the color-giving component), F (functional group that aids bonding), T (bridging group), and X (the reactive group that forms the covalent bond with the fiber). The reactive group — typically a triazine, pyrimidine, or vinyl sulfone system — is what distinguishes reactive dyes from every other dye class and drives their most significant operational challenge: hydrolysis.
Reactive dyes are primarily used on cellulosic fibers such as cotton, rayon, and flax, though polyamide, wool, silk, and acetate fibers can also be dyed using reactive dye chemistry. The dye is favored for achieving vibrant, long-lasting colors on natural textiles — particularly in hand dyeing yarn for beginners, where Procion MX cold-brand dyes fix at room temperature in a simple soda ash bath. However, the same reactive chemistry that creates the permanent bond also makes the dye vulnerable to hydrolysis: hydroxide ions in the alkaline dye bath compete with fiber hydroxyl groups, attacking the dye’s reactive group and forming hydrolyzed dye that cannot bond with the fiber. This hydrolysis side reaction reduces fixation efficiency by 10–30% in commercial operations, making it the central technical challenge of reactive dyeing.
The seven primary commercial reactive dye brands are Procion (ICI, UK), Novacron (Huntsman, Switzerland), Remazol (Hoechst, Germany), Levafix (Bayer, Germany), Reactone (Geigy, Switzerland), Primazin (BASF, Germany), and Drimarine (Sandoz, Switzerland).
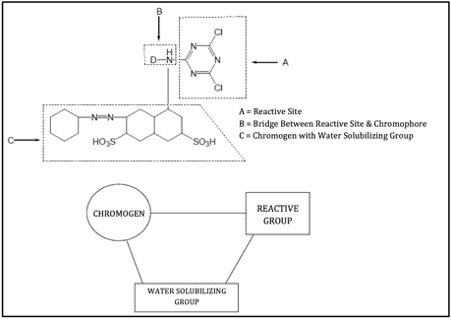
The components of reactive dye structure are:
- S = Solubilising groups (such as –SO₃Na or –COONa, or a combination of both)
- F = Chromophoric group, typically an azo, metal-complex azo, or anthraquinone system
- T = Bridging group that attaches the reactive system X to the chromogen F; commonly –NH–, –O–, –NHCO–, –OCH₃–, or –SO₃–
- X = Reactive system or group, which undergoes a chemical reaction with the functional group of the fibre
Here is a structural diagram showing the components of a reactive dye molecule:
Trade Names of Reactive Dyes
The reactive dye industry is dominated by several major manufacturers across Europe. Seven widely recognized trade names are listed below, representing the primary commercial brands used in textile coloration worldwide.
| Brand Name | Manufacturer | Country |
|---|---|---|
| Procion | I.C.I. | United Kingdom |
| Novacron | Huntsman | Switzerland |
| Remazol | Hoechst | Germany |
| Levafix | Bayer | Germany |
| Reactone | Geigy | Switzerland |
| Primazin | BASF | Germany |
| Drimarine | Sandoz | Switzerland |
Classification of Reactive Dyes
1. On the Basis of Reactive Group
Halogen-Based Reactive Groups
- Triazine group (e.g., Procion, Cibacron): Dichlorotriazine dyes activate at 30°C; monochlorotriazine dyes require 80°C for proper fixation
- Pyrimidine group (e.g., Reactone): Difluorochloropyrimidine systems activate at 40°C
- Quinoxaline group (e.g., Levafix): Dichloroquinoxaline derivatives fix at 40°C
Activated Vinyl Compound Reactive Groups
- Vinyl sulphone (e.g., Remazol): The active vinyl sulfone group forms under alkaline conditions at 40°C
- Vinyl acrylamide (e.g., Primazine): Fixes via Michael addition to fiber nucleophiles at 40°C
- Vinyl sulphonamide (e.g., Levafix): Contains a protected vinyl sulfone group that activates in the dye bath
2. On the Basis of Reactivity
- Low reactive dyes: pH is maintained at 12.0–12.5 using sodium hydroxide (NaOH) in the dye bath; fixation occurs over 60–90 minutes at temperatures up to 93°C
- Medium reactive dyes: pH is maintained at 11.0–12.0 using sodium carbonate (Na₂CO₃); typical dyeing temperature is 60–70°C
- High reactive dyes: pH is maintained at 10.0–11.0 using sodium bicarbonate (NaHCO₃); these cold-brand dyes fix at temperatures between 30°C and 60°C
3. On the Basis of Dyeing Temperature
Cold Brand Reactive Dyes
Cold brand reactive dyes contain highly reactive groups such as dichlorotriazine, enabling dyeing at temperatures between 32°C and 60°C. Examples include Procion M and Levafix E. These dyes are preferred for energy-efficient processes and sensitive fibers where lower temperatures prevent damage.
Medium Brand Reactive Dyes
Medium brand reactive dyes contain groups of moderate reactivity, requiring higher temperatures than cold brand dyes. Dyeing occurs at 60°C to 71°C. Remazol and Levafix are representative medium brand dyes. These provide a balance between energy consumption and fixation efficiency.
Hot Brand Reactive Dyes
Hot brand reactive dyes contain reactive groups of lowest reactivity, requiring temperatures of 72°C to 93°C for proper fixation. Procion H and Cibacron are hot brand dyes. The higher temperature overcomes the lower reactivity of the functional groups, ensuring complete covalent bond formation with the fiber.
Recent Classification of Reactive Dyes
The modern classification system for reactive dyes categorizes them by controllability mechanism rather than reactive group alone. Three distinct categories define current industrial practice:
| Type | Reactivity | Condition | Temperature | Reactive Group |
|---|---|---|---|---|
|
Salt Controllable |
Low reactivity in alkaline conditions |
Appreciable substantivity; careful salt addition is required |
80°C |
Monochloro Triazine, Monofluoro Triazine, Trichloro Pyrimidine |
|
Alkali Controllable |
High reactivity |
Moderate substantivity; careful alkali addition control is required |
Low temperature application (30–40°C) |
Dichloro Triazine, Difluorochloro Pyrimidine, Vinyl Sulfone |
|
Temperature Controllable |
Moderate reactivity |
Neutral conditions during application |
High temperature (80–98°C) |
Trichloropyrimidine and related groups |
Dyeing Mechanism of Reactive Dyes
The dyeing of textile material with reactive dyes occurs in three distinct stages: exhaustion of the dye onto the fiber, fixation via covalent bond formation under alkaline conditions, and wash-off of unfixed dye from the material surface. Each stage is critical to achieving level dyeing and optimum color fastness.

- Exhaustion: Dye absorption onto the fiber surface in the presence of an electrolyte
- Fixation: Covalent bond formation between the reactive group and fiber polymer under alkaline conditions
- Wash-off: Removal of unfixed and hydrolyzed dye from the material surface
Absorption of Reactive Dyes
When fiber is immersed in dye liquor, an electrolyte is added to promote dye exhaustion. Sodium chloride (NaCl) at concentrations of 20–80 g/L is typically used for cotton dyeing. The electrolyte neutralizes the negative surface charge of cotton fibers, reducing the zeta potential and enabling dye anions to approach and adsorb onto the fiber surface. After adsorption, dye molecules diffuse into the fiber interior, establishing equilibrium between exhausted and free dye in the bath.
Fixation of Reactive Dyes
Fixation is the chemical reaction between the reactive group of the dye and the terminal –OH groups of cellulosic fibers (or –NH₂ groups of polyamide and wool fibers), forming a strong covalent ether bond with the fiber polymer. This stage requires precise pH control through alkali addition. The alkali serves two functions: it ionizes fiber hydroxyl groups to nucleophilic fiber–O⁻ anions, and it activates the dye’s reactive group for nucleophilic attack. Two primary reaction mechanisms occur simultaneously during fixation:
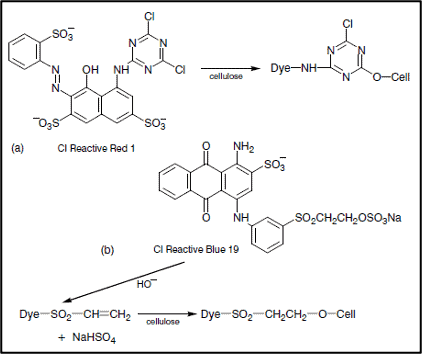
Types of Fixation Reaction
Two distinct chemical mechanisms govern reactive dye fixation on cellulose:
Nucleophilic Substitution Reaction
Cell–OH + HO⁻ → Cell–O⁻ + H₂O
Cell–O⁻ + Dye–Cl → Cell–O–Dye + Cl⁻
In halogen-substituted triazine dyes, the chloride atom is displaced by the fiber anion, forming a covalent ether bond between the dye chromophore and cellulose.
Nucleophilic Addition Reaction
Cell–O⁻ + Dye–SO₂–CH=CH₂ → Dye–SO₂–CH=CH₂–O–Cell
In vinyl sulfone dyes, the fiber anion adds across the activated vinyl double bond via Michael addition, creating the covalent dye-fiber bond. The vinyl sulfone group is often present in its protected β-sulfatoethyl sulfone form in the dye bath, converting to the active vinyl form under alkaline conditions.
Wash-off of Reactive Dyes
After dyeing is completed, a systematic wash-off sequence removes unfixed and hydrolyzed dye from the material surface. This step is essential for achieving level dyeing and high wash fastness. The wash-off procedure consists of three sequential stages: a hot water wash at 80–95°C to remove surface dye, a cold water rinse to cool the material, and a soap solution wash at 60–80°C with nonionic detergent to remove hydrolyzed dye trapped in the fiber. Insufficient wash-off results in poor rub fastness and reduced color fastness to washing.
Properties of Reactive Dyes
Reactive dyes exhibit specific performance characteristics that define their suitability for different textile applications:
- Available in powder, liquid, and print-paste forms to suit different application methods
- Fully water-soluble due to sulfonate (–SO₃Na) solubilizing groups
- Light fastness rating of 6 on the 8-step ISO blue wool scale, providing good resistance to ultraviolet-induced degradation
- Wash fastness rating of 4–5 due to the stable covalent bond between the dye reactive group and fiber polymer
- Perspiration fastness rating of 4–5, indicating resistance to color change under acidic perspiration conditions
- Produces brighter shades compared to direct dyes and sulfur dyes on cellulosic fibers
- Moderate rubbing fastness, improved by proper wash-off procedures after dyeing
- Moderate chlorine fastness; not recommended for applications involving exposure to hypochlorite bleaches
Dyeing Parameters for Reactive Dyes
Successful reactive dyeing requires precise control of multiple process parameters. The following factors must be carefully managed in commercial production:
| Parameter | Typical Range | Notes |
|---|---|---|
| pH of dye bath | 10.5–11.2 (alkaline) | Maintained by NaOH, Na₂CO₃, or NaHCO₃ depending on dye reactivity |
| Dyeing temperature | 30°C–93°C | Determined by dye brand (cold, medium, or hot brand) |
| Electrolyte (NaCl) concentration | 20–80 g/L | Salt concentration increases with shade depth |
| Dyeing time | 60–90 minutes | Includes exhaustion and fixation phases |
| Liquor ratio | 5:1–20:1 | Lower ratios improve dye bath efficiency but require tighter process control |
Reactive Dye Hydrolysis
Reactive dyes activate when dissolved in alkaline solution for cellulose dyeing. However, when dye solutions stand for extended periods, the dye loses reactivity. The primary cause is hydrolysis: hydroxide ions (OH⁻) from the alkaline bath compete with fiber hydroxyl groups as nucleophiles, reacting with the dye’s reactive group to form a hydrolyzed dye that cannot bond with the fiber.
The hydrolysis curve follows a two-phase pattern: dye concentration initially increases in the bath as the protective group separates, then decreases as the hydrolyzed dye becomes permanently detached from the reactive site. The concentration of hydroxyl compound rises continuously throughout hydrolysis, progressively reducing the dye’s ability to react with the fiber. Hydrolyzed dye remains in the bath and must be removed during wash-off; it contributes no color to the fabric and reduces overall fixation efficiency by 10–30% in typical commercial dyeing operations.
Stripping of Reactive Dyes
Reactive dyes cannot be fully stripped from fiber once the covalent bond has formed between the dye molecule and the fiber polymer. Complete stripping is chemically impossible due to the stability of the ether bond. Partial stripping becomes necessary when uneven dyeing or incorrect shade occurs, and involves breaking some dye-fiber bonds without destroying the fabric.
Partial Stripping Procedure
Partial stripping is achieved by treating dyed fabric with dilute acid at elevated temperature. The following formulations are standard in industrial practice:
- Glacial acetic acid: 5–10 mL per 1000 mL water bath; temperature raised to 70–100°C
- Formic acid: 2.5–10 mL per 1000 mL water bath; temperature maintained at 70–100°C until the desired shade reduction is achieved
The acid protonates fiber hydroxyl groups, breaking some dye-fiber covalent bonds. Process time varies by shade depth and target correction level. Multiple partial stripping cycles may be required for severe cases of uneven dyeing.
Advantages of Reactive Dyeing
- Produces brilliant, bright colours with high chromaticity on cellulosic fibers
- Permanent coloration through covalent fixation — the dye becomes part of the fiber polymer
- High wash fastness (rating 4–5) due to the covalent bond being resistant to repeated laundering
- Applicable across a wide temperature range from 30°C cold-brand systems to 93°C hot-brand processes, allowing energy optimization
- Bifunctional dyes offer improved fixation efficiency with multiple reactive groups
- Suitable for both exhaust dyeing and continuous padding applications
Disadvantages of Reactive Dyeing
- Incomplete fixation due to hydrolysis side reaction — typically 50–80% fixation efficiency in commercial dyeing
- Requires extensive wash-off procedures to remove hydrolyzed dye for high wash fastness
- High concentrations of electrolyte (20–80 g/L NaCl) are required for cotton exhaustion, increasing effluent salt load
- Reactive dye effluent is difficult to treat — azo dyes in wastewater can be reduced to aromatic amines under anaerobic conditions
- Many reactive dyes are not readily biodegradable; specialized effluent treatment is required
- Limited storage life — prepared dye solutions must be used within hours to prevent hydrolysis
Precautions for Reactive Dyeing
- Hot water must not be used for high-reactivity cold brand dyes — elevated temperature accelerates hydrolysis of the reactive group before fiber contact
- Prepared dye solutions cannot be stored for later use — hydrolysis begins immediately in alkaline solution and reduces reactivity within 2–4 hours
- Low-reactivity dyes are preferred for print paste formulation to prevent premature fixation during printing
- Inhalation of reactive dye dust poses health risks — a properly fitted N95 or higher-rated dust mask is required when handling powdered dyes
- Most reactive dyes have a maximum shelf life of 12 months in sealed containers; liquid dyes have shorter effective storage periods
- Alkali contact with skin and eyes must be avoided — appropriate PPE including gloves and safety glasses is mandatory
Temperature’s Effect on Fiber Reactive Dyeing Processes
Temperature is one of the most critical parameters in reactive dyeing, directly affecting both fixation efficiency and hydrolysis rate. Higher dyeing temperatures accelerate the diffusion of dye molecules into the fiber interior, promoting faster and more uniform color absorption. However, elevated temperatures simultaneously increase the rate of hydrolysis — the competing reaction where hydroxide ions attack the dye’s reactive group instead of the fiber. The optimal dyeing temperature is therefore a balance between fixation kinetics and hydrolysis control, and is determined by the specific reactive group chemistry of the dye being used. Cold brand dyes (30–40°C) minimize hydrolysis but require longer dyeing times, while hot brand dyes (80–93°C) achieve rapid fixation but require precise temperature control to prevent hydrolytic degradation of unfixed dye. For a comprehensive analysis of how temperature specifically affects fiber reactive dye performance, refer to our detailed guide on temperature’s impact on fiber reactive dyes.
Frequently Asked Questions
1. Why are bifunctional reactive dyes preferable?
Bifunctional reactive dyes contain more than one identical functional group within their molecular structure. This design provides a secondary fixation mechanism: if one functional group undergoes hydrolysis during the dyeing process, the remaining functional group(s) can still react with the fiber. This results in a higher overall degree of fixation compared to monofunctional dyes. Bifunctional dyes also offer greater tolerance for variations in process conditions, reducing the risk of unlevel dyeing.
2. Why were reactive dyes developed?
Reactive dyes were developed to overcome the fundamental weakness of direct dyes on cotton: poor wash fastness. Direct dyes rely on physical adsorption and hydrogen bonding to the fiber, which reverses during washing. The textile industry also found sulfur dyes and vat dyes too complicated for many applications. The 1956 introduction of dichlorotriazine reactive dyes by ICI (Stephen and Rattee) provided a practical solution — a dye that formed a permanent covalent bond with cellulose through a relatively simple exhaust dyeing process.
3. When were fiber reactive dyes first discovered?
During the early 1900s, chemists first proposed incorporating dyes with a covalent bond directly into the fiber polymer. In 1955, Stephen and Rattee at Imperial Chemical Industries (ICI) in England successfully demonstrated the first practical reactive dyeing of cotton using a dichlorotriazine-containing dye. This breakthrough led to the commercial introduction of the Procion dye series, launching the modern reactive dye industry.
4. What is dye hydrolysis in reactive dyeing?
Dye hydrolysis is the primary cause of incomplete fixation in reactive dyeing. Under alkaline conditions, hydroxide ions (OH⁻) compete with fiber hydroxyl groups as nucleophiles, attacking the dye’s reactive group. This produces hydrolyzed dye — a dye molecule that has reacted with water rather than the fiber. Hydrolyzed dye cannot form a covalent bond with the fiber, remains in the bath, and must be removed during wash-off. Hydrolysis reduces fixation efficiency by 10–30% in commercial dyeing, making it one of the most significant technical challenges in reactive dyeing.
5. Are reactive dyes colorfast?
Reactive dyes provide good to excellent colorfastness properties when properly applied and wash-off is complete. Wash fastness reaches rating 4–5 (ISO 105-C03) due to the covalent bond resisting removal during laundering. Light fastness is rated 6 on the 8-step ISO blue wool scale. Peroxide bleaching and chlorine exposure present moderate challenges — certain chromophores (particularly azo-based dyes) are susceptible to reductive cleavage under these conditions.
6. Can fiber reactive dyes be used for tie-dye?
Fiber reactive dyes are the standard choice for tie-dye and other fiber arts applications on natural fibers. Procion MX dyes (cold brand) are particularly popular for tie-dyeing because they fix at room temperature (20–25°C) in an alkaline soda ash bath, making them safe and practical for craft use. The dye forms permanent covalent bonds with cotton, rayon, and other cellulosic fibers, producing color that withstands repeated machine washing without fading.
References
- Royal Society of Chemistry. (n.d.). Reactive Dyes. In RSC Chemistry World. https://en.wikipedia.org/wiki/Reactive_dye
- Royal Society of Chemistry. (n.d.). Dye. In RSC Chemistry World. https://en.wikipedia.org/wiki/Dye
- Society of Dyers and Colourists. (1995). Cellulosics Dyeing. Bradford, West Yorkshire: SDC.
- Broadbent, A. D. (2001). Basic Principles of Textile Coloration. Bradford, West Yorkshire: Society of Dyers and Colourists.
- Shore, J., & Society of Dyers and Colourists (Eds.). (2002). Colorants and Auxiliaries: Organic Chemistry and Application Properties, Vol. 1: Colorants (2nd ed.). Bradford, West Yorkshire: SDC.
- Chakraborty, J. N. (2015). Fundamentals and Practices in Colouration of Textiles. WPI Publishing.
- Mahapatra, N. N. (2016). Textile Dyes. Woodhead Publishing India.

